Expanding Business and Capital Alliance Contract with HEALIOS K.K. to develop practical applications for regenerative medicine
July 10, 2019
TOKYO - Nikon Corporation (Nikon) agreed on the expansion of partnership between HEALIOS K.K. (Hardy TS Kagimoto, CEO, Tokyo, Japan, hereafter referred to as Healios) based on the business and capital alliance agreement concluded in 2017.
Based on this agreement, Nikon will accept 4 billion yen of series 2 unsecured convertible bonds with stock acquisition rights through a third-party allocation by Healios, and expand the business partnership. Nikon, as Healios' main partner, will advance Healios' development of practical applications for regenerative medicine. Nikon will contribute to this effort towards the practical use of regenerative medicine in Japan and to the improvement of QOL (quality of life) of people by leveraging the know-how and technologies of both companies.
About Healios
Regenerative medicine is a field that is expected to offer a new form of treatment for patients suffering from diseases without effective treatments worldwide, and with the potential to become a large market in the near future as efforts towards product development and practical applications become full-fledged. Healios is a front runner in the development of regenerative medicine using iPSCs (induced pluripotent stem cells), etc., and is a biotechnology company that possesses a number of seeds/products with the potential for practical applications.
Healios, established in 2011 with an initial public offering in 2015 (TSE Mothers: 4593), is promoting the research and development of regenerative medicine towards practical applications. In Japan, Healios has been conducting a clinical trial for ischemic stroke with a stem cell product since 2017, and in addition, has started a clinical trial for ARDS (Acute Respiratory Distress Syndrome) with the same product in April 2019. Healios is also conducting research and development of regenerative medicine products by using iPSC technology to create functional human organs in the body via organ bud transplantation, and is engaging in the development of various other new products.
(For details, please visit https://www.healios.co.jp/en/)
Background of partnership
Nikon accepted a 500 million yen third-party allocation of shares by Healios (previously: Retina Institute Japan, K.K.) in August 2013 and has supported Healios' development of regenerative medicine targeting age-related macular degeneration using iPSCs. Based on the business and capital alliance agreement concluded in February 2017, Nikon has accepted a 2 billion yen third-party allocation of shares by Healios, and has advanced the development of practical applications for regenerative medicine by leveraging the strengths of both companies.
Purpose of this agreement
Nikon, using its advanced optics and imaging analysis technologies, has provided solutions such as the comprehensive cell quality evaluation system and drug discovery support service. These solutions have been adopted in a variety of fields. Nikon CeLL innovation Co., Ltd. (Nikon CeLL innovation), a wholly-owned subsidiary of Nikon, is providing a wide range of process developments and manufacturing services, from pre-clinical to commercial manufacturing of cell and gene therapies.
Since 2017, Nikon CeLL innovation, in collaboration with Athersys, Inc. (Gil Van Bokkelen, Chairman and Chief Executive Officer, United States, hereafter referred to as Athersys) and Healios, has been providing manufacturing support for commercialization of MultiStem® for ischemic stroke in Japan.
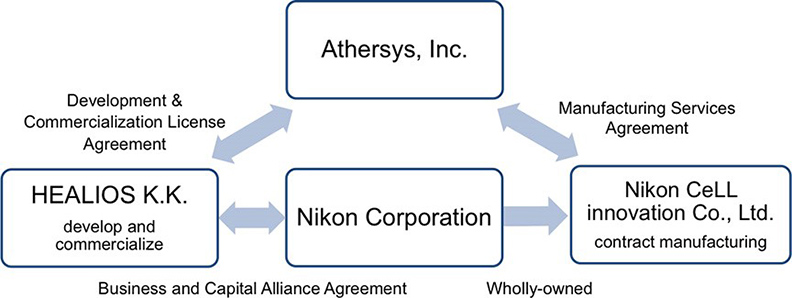
MultiStem is an adult-derived stem cell product suitable for mass production developed by Athersys. Currently, several clinical trials using MultiStem are underway for various indications, including cardiovascular, neurological and inflammatory/immune conditions. Athersys holds the patent and licensing rights to this important technology, and has conducted multiple studies demonstrating that it exhibits a consistent safety profile.
Healios has started two clinical trials for ischemic stroke and ARDS making use of MultiStem in Japan. Nikon CeLL innovation has conducted activities based on the manufacturing services agreement with Athersys and will advance to prepare for the growing demand for commercialization based on this agreement as of today. In addition, Nikon CeLL innovation will enter into discussion and negotiations with Healios to establish the contract development and manufacturing for multiple products developed by Healios.
Implications for Nikon Group business
By expanding the partnership based on the business and capital alliance agreement, Nikon Group will have a wide range of business opportunities in the contract manufacturing of cells for regenerative medicine.
If Healios acquires the license to develop regenerative medicine products for Japan, Nikon Group has exclusive negotiation rights or an opportunity for preferential negotiation (excluding specific fields) in the case where Healios considers contract manufacturing of cell production for regenerative medicine. In addition, Nikon will develop useful information from Healios concerning areas such as cell contract manufacturing and image evaluation which can contribute to the development of new businesses in the field of regenerative medicine.
By signing this agreement, Nikon Group and Healios will promote the establishment of new treatments for diseases currently without effective treatments by leveraging the technology and knowledge of both companies. Furthermore, we will develop practical applications for regenerative medicine and participate in the expansion of related industries.
About Acute Respiratory Distress Syndrome
Acute Respiratory Distress Syndrome (ARDS) is a general term for the symptoms of acute respiratory failure suddenly occurring in seriously ill patients. The major causes are severe pneumonia, septicemia, trauma etc. Inflammatory cells are activated in response to these diseases or injuries, causing damage to the tissue of the lungs. As a result, water accumulates in the lungs, leading to acute respiratory failure. According the ARDS treatment guideline 2016, the mortality rate is approximately 30 to 58%.
Artificial respiration using an endotracheal tube or mask is used to treat respiratory failure in an intensive care unit. However, it is known that prolonged use of a ventilator worsens a patient's prognosis. There is demand for a new treatment for ARDS that will lead to improvement in patients' symptoms and prognosis.
- *MultiStem® is a registered trademark or trade name of Athersys, Inc. in the US and other countries.
The information is current as of the date of publication. It is subject to change without notice.
